Cycles
Like in all ecosystems, biological cycles will occur in your tank. These cycles help organisms live in happy cohabitation!
Why are cycles important?
Cycles
are important because they maintain homeostasis in a system. Homeostasis is the
ability for an organism or system to maintain internal equilibrium. In your
tank you have chemical and biological equilibriums.
What cycles are in my tank?
The cycles in your tank include:
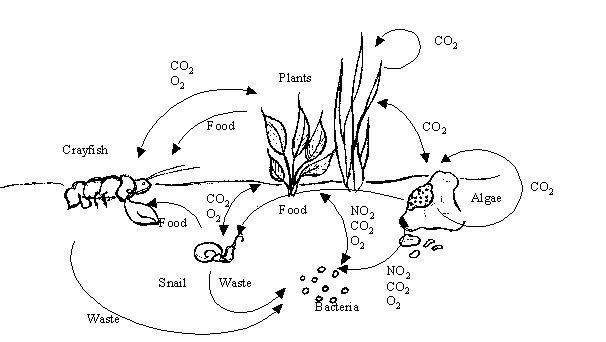
The above picture is a representation of all the major cycles that are occurring within the crayfish environment. For instance, the food chain is exhibited, with the arrow pointing to the crayfish from the plant and the snail. The photosynthesis and respiration cycles are represented by the plants taking in the carbon dioxide produced by the organisms and the organisms consuming the oxygen produced by the plants. The nitrogen cycle is represented by the snail and the crayfish giving off waste, which is consumed by the bacteria and turned into NO2 which is consumed by the plants.
back to the top

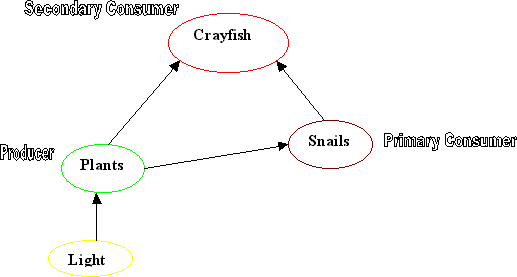
The Food Web
Food webs occur in all natural environments and
describe the interaction of the plants and animals living in the ecosystem.
They show how food and energy move between them. At least three levels exist in
all food webs including producers, primary consumers and secondary consumers.
More levels can exist as the web becomes more complex from the addition of
higher consumers.
Producers
Producers
are organisms that get their energy from sunlight and convert it to useable
energy via photosynthesis. In your tank, the producers include the plants and
algae.
Primary Consumers
Primary
consumers get their energy by consuming the producers and no other consumers.
They are strictly vegetarians! Snails and any other small plant eating animals
in your tank make up this group.
Secondary Consumers
Secondary
consumers get their energy from consuming both producers and primary consumers.
Like us, they like to eat both meat and plants. The crayfish are your secondary
consumers because they eat plants, algae, small animals and sometimes even each
other!
back to the top

Photosynthesis & Respiration
Photosynthesis is the light induced process by which green plants produce oxygen from carbon dioxide.
In the aquarium, carbon dioxide produced by the crayfish and aerobic bacteria,
is dissolved in the water. During
photosynthesis, the carbon dioxide is taken inside the plant and converted into
simple sugars. This synthesis reaction can be written chemically in the
equation:
6CO2 + 6H2O  C6H12O6 + 6O2 C6H12O6 + 6O2
Carbon
Water
Sugar Oxygen
Dioxide
Several factors influence the rate at which photosynthesis occurs in aquatic plants. The rate increases as the light intensity increases, as the water
temperature increases, and in the presence of high concentrations of carbon
dioxide. It is important to select plants that will survive within the aquarium
temperature and light constraints.
Respiration
is the process by which plants and animals convert the energy stored in food
into useable forms of energy, such as heat. Living cells in aquatic organisms
use the dissolved oxygen in the water to convert sugars into heat. Chemically,
respiration is the reverse of photosynthesis
C6H12O6 + 6O2  6CO2 + 6H2O 6CO2 + 6H2O
Sugar + Oxygen Carbon Dioxide & Water
back to the top

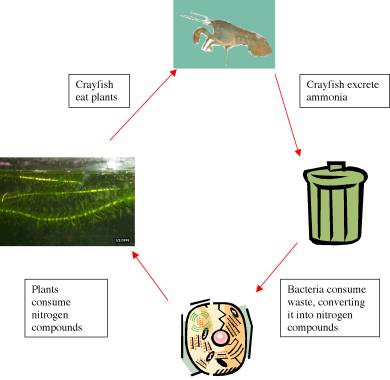
The Nitrogen Cycle
Nitrogen exists in the environment, and your tank, in several forms and is continuously
converted between these forms via the nitrogen cycle. These forms are: nitrate
(NO3), nitrite (NO2) and ammonia (NH3).
A simple breakdown of the cycle is:
- Plant and animal wastes and dead plants and animals decompose adding nitrogen to the soil (or water) as ammonia.
- The bacteria in the soil (or on the gravel) then convert these forms to forms that plants can use.
- The plants then use the nitrogen to grow.
- Animals (the crayfish and snails) eat the plants and the plant and animal residues return to the soil (or water) again completing the cycle.
Ammonia is toxic to the crayfish at high concentrations so it is important that it never reaches high levels. That is why it is so important to have a good bacterial population in your tank. The bacteria will convert the ammonia into nitrite and then to nitrate thereby reducing it's toxicity by a process called
nitrification.
What is nitrification?
Nitrification involves two bacterial species: Nitrosospira bacteria, which oxidizes ammonia to
nitrite, a slightly less toxic form of Nitrogen, and Nitrobacter bacteria, which converts nitrite to nitrate,
a form of nitrogen that is much less toxic then either of the other two forms.
Both species utilize energy released by the reactions. This complex series of
reactions can be summarized in chemist shorthand as:
Nitrosospira:
55NH4++
76 O2 + 109HCO3-ŕ C5H7O2N + 54NO2-+
57H2O + 104H2CO3
Nitrobacter
400NO2-
+ NH4+ + 4H2CO3 + HCO3-
+ 195 O2 ŕ C5H7O2N + 3H2O + 400
NO3
How do I know if my bacterial populations are healthy?
Nitrate, nitrate, ammonia and dissolved oxygen levels are good indicators of
how the bacterial processes are functioning. Since nitrite is highly toxic when
in an ecosystem its ideal level remains approximately at 4 mg/L. Nitrate levels are not as critical, and
are ideally no higher than 60 mg/L. Ammonia levels should not rise above 2
mg/L. Since both of the bacteria use oxygen in this process a good level of
dissolved oxygen is needed. Dissolved oxygen levels around seven or eight mg/L
are ideal, and levels at or below two are cause for concern.
A simplified sketch of the nitrogen cycle is shown here:
back to the top
|